Meet Maria, the game-changer
Solar superhero
Call them superheroes.
The crystals that caught her eye six years ago probably would not not seem special to an untrained eye. But tweak them a little – add a little something to them, like an organic molecule – and suddenly these simple, robust semi-conductors become capable of so much, they just might change the world.
To Maria Antonietta Loi, professor of Photophysics and Optoelectronics at UG, the crystals are like Pegasus, a mythological creature that had properties of both horse and bird. Or, like Spiderman – to modernize the analogy. ‘These are hybrids too. The fact that they combine attributes from different origins gives them amazing properties.’
Perovskite
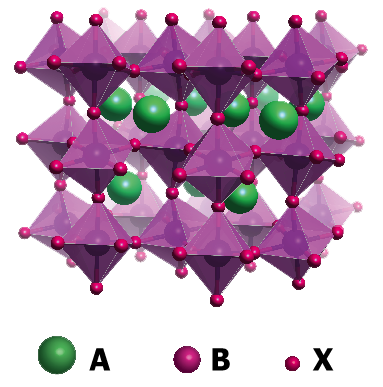
A perovskite is a crystal that was first discovered in 1839 and named after the Russian minerologist Lev Perovski. These days perovskites form a large family of crystals where the atoms composing them are positioned like a cube, or a pseudocube.
To create a hybrid perovskite you have to juggle the different components. In the centre (B) of a octahedron you’ll find metals: lead, or tin. On the corners (X) of the octahedron there’s usually a halogen ion, like chloride, or Iodine. The octahedron is inside a cube that has corners (A) of organic kations, like methylammonium or formadinium.
Changing the elements of the respective positions changes the properties of the crystal.
She’s talking about perovskites. And not just any perovskite, but her own, which she puts together in her lab. She tweaks them, by combining organic and anorganic components. She mixes them in the clean room, where absolutely no dust or any other contaminate is allowed. She spreads them out in gloveboxes, careful not to expose them to the negative effects of water or oxygen. She excites them, by exposing them to bright laser lights. She measures them, by putting them under a tiny, artificial sun.
Above all, she seeks to understand them. Maybe they will give away their secrets – secrets she could put to use.
Amazing discoveries
And she’s well on her way to uncovering the secrets of the perovskite crystals. In the last six years, Loi has made some amazing discoveries. One of her hybrids turned out to be extremely sensitive to X-rays. It has since been used to make an X-ray detector four times more efficient than regular ones. That means an X-ray that doesn’t need use as much radiation to detect, say, a broken bone – and therefore does less damage to the body.
But the real sensation? The perovskites she uses to build solar cells. These aren’t just any solar cells: they promise huge levels of efficiency. And that’s only one part of the research that recently earned her the Physics Prize 2018 – a prize that has also been awarded to the likes of Nobel laureates Gerard ’t Hooft and Gerard Veltman, as well as the famous physicist Robbert Dijkgraaf.
Regular silicon solar cells have a maximum efficiency of about 25 per cent. Perovskite solar cells are closing in on that threshold quickly: they have already reached an efficiency of 22 percent. That may be less efficient than old school cells, Loi says, but ‘but you have to remember that that number was reached in seven years of research, while it took forty years to get silicon there.’ And there’s potential for more: up to a theoretical maximum of 33 percent efficiency.
Hot electron
Why? Because perovskites absorb lots of light and conduct electrical currents well. They can also be dissolved easily in a liquid and grown into flexible layers that you might even put on your clothes one day. They are robust to defects, resilient. And best of all, they’re cheap.
However, there are problems with the crystals. For example, regular perovskite solar cells contain lead, a very toxic metal that just isn’t suitable for mass production. And unfortunately, just like with silicon cells, a lot of the solar photon energy leaks away. A photon has to ‘hit’ a solar cell with just the right amount of energy; when it has too much, it produces ‘hot’ electrons that make crystals of a solar cell vibrate and heat up, and the energy is lost.
‘We were studying a tin-based perovskite to see if we could do without lead; I wanted to study the basic properties of this perovskite’, she says. She wants to know exactly what makes the perovskite tick.
And then, Loi stumbled upon a special type of perovskite, one that might change everything. Together with her post doc Hong-Hua Fang, she experimented with a hybrid that contained tin instead of lead. Tin is a lot less toxic, and would she hoped it would help them find a way to solve the toxicity problem. The tin hybrid got great results in solar cells, too. The solar cells made by the other post doc, Shuyan Shao, had an efficiency of 9 percent: a record for a hybrid perovskites without lead.
Thousand-millionths of a second
‘But then we found something amazing’, she says. ‘Somehow, the crystal vibrated far less than usual. In this particular perovskite the hot electrons kept their energy for a very long time.’
A long time – really, a few nanoseconds, instead of the few hundred femtoseconds Loi was used to. A femtosecond is one millionth of one billionth of a second. ‘So that is a thousand times slower than usual’, she explains.
And even though to any regular observer the energy is still lost very quickly, to Loi the slow-down opens up a vast array of possibilities. Those few nanoseconds might be enough to get the energy out of the material and transfer it somewhere else, where it can be put to use. This could potentially increase the efficiency to as much as 66 percent – a game changer.
I always look for new things that I don’t understand, but that are interesting and promising, so I don’t get bored
Of course, she’s not there yet. Now she will have to accomplish two things: first, she must find out why the tin in the crystal makes the hot electrons slow down. Second, she must find a way to extract the energy.
But she has some ideas about how to do that. And that’s where her enquiring, restless mind comes in handy. ‘I couldn’t do the same thing for thirty years. I always look for new things that I don’t understand, but that are interesting and promising, so I don’t get bored.’
Superatom
Before she started researching perovskites six years ago, Loi worked on nanotubes. It’s an on-going project; recently she succeed in making nanotubes arrange themselves in a substrate. She’s also worked on another hybrid: quantumdots, which are clusters of a thousand atoms that behave like one single super-atom. These quantumdots, laid out in a paintlike film, can turn light into electricity. Loi’s group recently succeeded in improving the quantumdots and adjusting the electronic properties.
Having all these ideas and materials at hand has proven to be very useful for her current project. Loi thinks she can do something special with the quantumdots: ‘They might make for a very interesting material to try and get the energy out of the perovskite solar cells’, she says.
And if that doesn’t work out, she will no doubt find another way. ‘To my brain it’s all the same’, she says. ‘I have a holistic approach to science. I just look around to see what I can use. I just want to do beautiful research.’